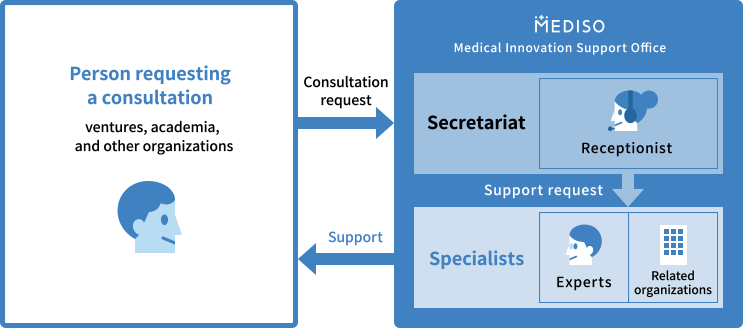
Specialists Introduction
Medical Innovation Support Office support consultants in cooperation with experts (specialists) in each field of R&D, pharmaceutical affairs and insurance, intellectual property management, management and financial accounting, marketing, legal affairs, and international development, as well as related organizations, including the Ministry of Health, Labour and Welfare.
What is Specialists
Experts in each field of R&D, pharmaceutical affairs and insurance, intellectual property management, management and financial accounting, marketing, legal affairs, and international development.
Depending on the specifics of your request, we offer support in cooperation with appropriate specialists and related organizations, including the Ministry of Health, Labour and Welfare.
Introduction of Support Providers
We will introduce registered support providers
(updated as necessary).
Search the area
Search the area
Kazuhiro Ikumi
MEDICOLAB Co., Ltd.
Main specialty areas
- Medical devices
Specialized support fields
- Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management - Other
Board-certified neurologist, CEO at MEDICOLAB, Co., Ltd. My initial job was a medical doctor at a tertiary care emergency hospital. I have 14 years of clinical experience in Parkinson's disease, dementia and other neurodegenerative diseases. MEDICOLAB, Co., Ltd. receives PR and development support from Fujitsu, NVIDIA, Amazon, and Microsoft. I am involved in business development in the CNS area and support researchers for their idea and trial products or services for commercialization.

Yutaka Kono
Guest professor
Drug Discovery Science Division,Institute for Open and Transdisciplinary Research Initiatives, Osaka University
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Business
planning - Fundraising
- Management
Strategy - Other

Masaho Ishino
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Management
Strategy - Intellectual
property
management - Other
After engaging in medical research for 25 years, I became a patent attorney (JAPAN). Over the next 20 years, I have supported medical researchers in creating intellectual property and planning research strategies, as well as engaged in contract practice and technology transfer of university IP. Intellectual property strategies for regenerative medicine and other new modalities require not only a high level of expertise in both cutting-edge medical research and intellectual property rights, but also comprehensive knowledge covering clinical development and regulations. Utilizing my experience of contributing to the development research and practical application of university-originated seeds as a representative of an institution responsible for the AMED's Project of Translational and Clinical Research Core Centers and an organizer of nationwide academia intellectual property networks, I aim to contribute to raising the potential of intellectual property of academia/venture.

Takeshi komatani
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy - Intellectual
property
management - Other
Takeshi S Komatani is currently principal litigation certified patent attorney in the field of chemical/pharma and bio/life sciences at a patent law firm, and has a number of counsels, including in academia, start-ups and big pharma internationally. Takeshi received his PhD from the University of Tokyo, Japan, received his LLB and LLM (global legal practice) from Keio University, and is currently a visiting professor at Kobe University, teaching IP strategy in entrepreneurship and Doshisha University (Kyoto). He has completed CEIPI Summer School on IP (University of Strasbourg), France. He is also qualified as a pharmacist with a certificate of Kampo and natural medicines specialist qualification. He was a researcher at F Hoffmann-La Roche in Basle, Switzerland. He has completed the EU-recognised PharmaTrain course at Osaka University, and is qualified as a board-certified member of the Japanese Association of Pharmaceutical Medicine. He is a member of the AIPPI and vice chair of TRIPS SC and a member of IP-GRTK SC and Pharma SC, and a member of the editorial board of the Pharmaceutical Patent Analyst (UK). He is also a member of JPAA, APAA, IPAJ, PSA, JPA, CSAIP and AAAS. He has lectured in academia, including at Japanese and German universities and WIPO Academy, and contributed to a number of articles.

Jun Yamadera
Eyes, JAPAN co. ltd.
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy - Other
Jun Yamadera founded Eyes, JAPAN co. ltd. in 1995, the first IT startup from the University of Aizu, Fukushima. In the past 20 years, he has been working on various cutting edge projects such as exporting Fukushima rice via web in 1995, making world’s first virtual pottery system, making CG of historical archives of national treasure of Japanese castles, temples, traditional dances and medical motions using motion capture. He is a pioneer in Augmented Reality wearable technologies, Medical x IT and has been organizing world’s first medical security hackathon since 2012. His team won the the championship in Developers Challenge 2013 Health 2.0 in Silicon Valley. He is the Health 2.0 Fukushima Chapter Leader and a TEDxKobe 2015 Speaker. He started a project called “FUKUSHIMA Wheel” in the aftermath of the terrible disaster caused by the earthquake and nuclear accident in Fukushima, JAPAN in March 11, 2011. More than 24 years experience in Cutting Edge Technologies such as AI, Blockchain and Cyber Security. He is also advisor of Safecast and LivingAnywhere.

Kazushi Kamitani
JPRO INCUBATION OFFICE, FOUNDER
Main specialty areas
- Medicines
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management - Other
He worked for domestic & international life science firms in the areas of product development, marketing, PR&IR, international business, and business development.
After serving the US subsidiary of a Japanese firm as a board director & CFO and the European startup as CEO, he established the incubation office.
He supports oversea startups to explore Japan market from business & administration side, providing info, advisory and functional services.
International MBA, Certified Specialist of Intellectual Property Management, Financial Planner, IT Coordinator

Daisuke Sugiyama
Hiroshima University, Translational Research Center, Professor
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Fundraising
- Management
Strategy - Intellectual
property
management - Other
He obtained his PhD thesis at University of Tokyo. After working at several hospitals as a clinician, he has engaged in medical reserach at University Paris 6 (French Government Scholar), Dartmouth College (JSPS research fellow), Kyushu University and Hiroshima University. Based on the experiences working at ARO research core hospital and starting up bio-venture company, he supports and conducts both translational and clinical researches.

Hitoshi Fujimaki
TMI Associates Attorney-at-law (admitted in Japan and New York)
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Other
Joined TMI Associates in 2016. Completed an LL.M. in National and Global Health Law at Georgetown University Law Center in 2023, earning the Food & Drug Law Certificate. Admitted to the New York State Bar in 2024. The practice focuses on pharmaceutical, medical device, and healthcare regulatory matters in both Japan and the United States, with extensive experience supporting R&D and commercial operations of Japanese companies domestically and abroad. Work includes licensing and collaboration agreements with overseas partners, joint research and development arrangements, CRO agreements, GCP compliance during clinical trials, promotional review, regulatory assessments (including medical-device classification), authority inspections, and broad advisories on pharmaceutical and medical-device regulations. Also advises on U.S. healthcare compliance issues, including the Anti-Kickback Statute, the Sunshine Act, and related federal and state requirements. Author of Introduction to U.S. FDA Drug and Medical Device Law and Regulation (Shojihomu, September 2024).

Satoshi Ogawa
TMI Associates Partner (Attorney)
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Fundraising
- Management
Strategy - Intellectual
property
management - Other
Satoshi Ogawa, Ph.D., is a partner at TMI Associates specializing in life sciences and healthcare. After earning his Ph.D. in Life Sciences from Kyoto University, he joined TMI Associates as an attorney. Following a three-year secondment at a law firm in New Delhi, India, he has been based at TMI’s Kyoto Office since 2019. Dr. Ogawa advises both Japanese and international clients on cutting-edge technologies, including regenerative medicine, digital health, and AI-driven healthcare solutions. His work covers startup support, patent licensing, industry–academia collaboration, regulatory compliance, and intellectual property disputes. He works extensively with universities, startups, pharmaceutical and medical device companies, venture capital firms, and public institutions. He also serves as an advisor to BioCommunity Kansai (BiocK) and HVC KYOTO, and contributes to conflict-of-interest (COI) committees and institutional review boards (IRBs). Dr. Ogawa is passionate about advancing innovation and helping clients navigate complex legal and regulatory challenges in the life science ecosystem. His publications include “Startup Intellectual Property and Legal Guidebook – Key Points for Early-Stage Bio and Life Science Ventures” (METI-Kansai) and “Healthcare Business Legal Consultation Handbook” (Chuokeizai-sha).

Tetsuo Miura
Specially Appointed Researcher, Center for Clinical Sciences, National Center for Global Health and Medicine
Main specialty areas
- Medicines
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Management
Strategy - Other
I worked as a product and business development manager primarily in the fields of diagnostic equipment-reagent systems and medical devices at major foreign pharmaceutical and domestic medical device companies, and also participated in global product development teams at the US and French headquarters. I have medical science knowledge in the fields of infectious diseases, immunology, cardiovascular diseases, metabolism, reproductive medicine, and laboratory medicine, as well as information on the medical field. After mandatory retirement, I served as a senior researcher at the National Center for Global Health and Medicine, responsible for developing and implementing international collaborative clinical research and clinical trials for pharmaceuticals and medical devices with clinical trial networks in Southeast Asian countries. Currently, as part of an industrial-medical collaboration project, I also provide guidance and support to companies in product development with an eye toward exit strategies such as obtaining pharmaceutical or WHO PQ approval, and being included in guidelines in cooperation with related medical sectors and societies.

Tomoyo Mori
Fast Track Initiative, Inc. Capitalist
Main specialty areas
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy - Other
Tomoyo obtained her medical license and began her career by engaging in training and clinical practice in medically underserved areas. She then joined the Department of Cardiovascular Medicine at Tokyo Women's Medical University Hospital, where she treated patients with heart failure, heart transplants, and myocardial infarctions. At the university hospital, she also participated in clinical trials for medical device startups. In parallel with her clinical work, she was involved in domestic and international business development for medical device startups and led a team while participating in U.S.-based MedTech accelerators (MedTech Innovator, Fogarty Innovation). She holds an MBA from the University of Michigan's Ross School of Business and is a board-certified internist by the Japanese Medical Specialty Board as well as a certified occupational physician by the Japan Medical Association. Currently, she is part of Fast Track Initiative, where she focuses on investment, company creation, and business development for medical device startups.

Hirohumi Shido
Main specialty areas
- Medical devices
Specialized support fields
- Other
I am a UI/UX design expert and specialize in driving product development. As a product manager at a medical startup, he was in charge of renewal and new planning for multiple SaMD products. My experience ranges from planning to obtaining regulatory approval, and I have won a Good Design Award for a healthcare product. These experiences have made me realize the importance of experience design and usability in SaMD products with a diverse mix of stakeholders.

Kazuhiro Terashima
CaTe inc. CEO
Fujita Health University, Department of Cardiology, Assistant Professor
Main specialty areas
- Medical devices
Specialized support fields
- Business
planning - Fundraising
- Management
Strategy - Other
Cardiologist, Fellow of Cardiovascular Intervention and Therapeutics, Registered Cardiac Rehabilitation Instructor, Certified Occupational Health Physician and Clinical Education Instructor. He graduated from Nagoya University School of Medicine in 2011. After working at the Japanese Red Cross Aichi Medical Center, Nagoya Daiichi Hospital, Sakakibara Memorial Hospital and other institutions, he joined Fujita Health University in 2023 as Assistant Professor of Cardiology and CCU Instructor. In 2020, he founded CaTe Inc. to research and develop programmed medical devices for cardiac rehabilitation, and to conduct clinical research and company-led clinical trials of such devices. He is currently the principal investigator for basic research on the Exercise Load Optimization function, a promising technology seed. In 2023 he closed Series A funding with JAFCO etc. as lead investor. While continuing his clinical work, he serves as CEO of the company and advises and supports other clinicians in the social implementation of clinical technology seeds.

Minori Kosuge
Senior Manager, Regulatory Affairs and Quality Assurance
Rokken, Inc.
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Other
- Experience of working for 15 years at a Japanese medical device company: Technical writing, regulatory affairs (US 510k submissions, translation for QSR inspection), R&D of patient monitor and its related programs.
- Experience of working at Japanese SaMD startup companies: experience of negotiation with PMDA and consultation for regulatory affairs/quality assurance.
- Currently working as a regulatory affairs/quality assurance consultant and an in-house regulatory affairs/quality assurance specialist.
Support available for class II active medical devices and SaMD, for Japan and US.

Mineki Takechi
Cardio Intelligence Inc. Chief Development Officer
Director
Main specialty areas
- Medical devices
Specialized support fields
- Business
planning - Management
Strategy - Other
After working at Fujitsu Ltd. on the development of products and services such as image processing system and cloud information service for home healthcare, as well as research and development in information retrieval and AI, and new business development, I engaged in the construction of a pulmonary hypertension patient registry at the International University of Health and Welfare Mita Hospital. Since 2020, I have been serving as CDO at Cardio Intelligence Inc., responsible for the development of software medical devices for arrhythmia analysis and regulatory affairs. Since 2021, I have also been serving as COO, overseeing overall business planning and execution.

keiji Asada
Amenichi Consulting, LLC, CEO
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy - Other
Graduated from Auburn University with a degree in Chemical Engineering. Currently, serving as the CEO of Amenichi Consulting, LLC, a consulting firm specializing in regulatory compliance (FDA) and marketing support for companies entering the U.S. market, particularly in the fields of pharmaceuticals, medical devices, and regenerative medicine products. With extensive industry experience and expertise, he assists clients in developing business plans, securing funding, and establishing operational structures. Additionally, he provides comprehensive support, from the formulation to the execution of business strategies, to help clients enhance their competitiveness. Furthermore, he leverages his network to match clients with the most suitable business partners. With a global perspective, he offers customized services tailored to the needs of each client, aiming for sustained growth and success. Amenichi was established in 2018 and has supported over 50 companies since its inception.

Ruka Takeuchi
Discovery Biotech Consulting Representative
PARKS Commercialization Producer
Main specialty areas
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Management
Strategy - Other
I have been engaged in marketing and business development for more than 30 years in the life science market, mainly in the medical and pharmaceutical fields, both in Japan and overseas. Currently, I have established my own consulting firm and provide business consulting services to foreign and domestic companies from start-ups to small and medium-sized enterprises. I would be happy to be of service to you by drawing on my past business experience as well as my daily experience in providing hands-on support to various companies and projects.

Teppei Konishi
Biomy Inc. CEO
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Business
planning - Fundraising
- Management
Strategy - Other
He completed his graduate studies at the Graduate School of Engineering Science, Osaka University. At NTT DOCOMO, he specialized in big data analysis and AI-driven video analysis research, contributing to new business development. After leaving NTT DOCOMO, he served as CTO at an IT venture before founding Biomy Co., Ltd. There, he led the development of a personalized medical platform utilizing AI technology, collaborating with domestic and international pharmaceutical companies and research institutions. He also secured a Class II medical device manufacturing and sales license.

Yuki Yamakage
Representative Director, Takashi Shimada Office Co., Ltd.
Managing Partner, BD Consulting Ltd.
Main specialty areas
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Other
I have experience working for a global medical device company in insurance strategy development and insurance applications for products in a wide range of medical specialties and disease areas, as well as market access work (relevant academic societies, patient groups, government agencies and industry associations). I have now set up my own private practice and provide support to medical device and pharmaceutical companies and new entrants in terms of healthcare and care systems and insurance strategies. I have experience in the insurance application category (C1/C2 applications) for innovative medical devices. I also have a lot of experience in training employees and conducting seminars, most recently on SaMD insurance applications. My motto is to provide consulting services that address the concerns of my clients after explaining information on insurance applications for medical devices in a way that is easy to understand for beginners.

Tomohisa Hayasaki
GVA LPC Attorney-at-Law / Partner
The Leader of The Team ""Medical / Healthcare / Cosmetic Industry""
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Fundraising
- Intellectual
property
management - Other
After registering as a lawyer, I have been handling disputes such as corporate and labor disputes, as well as providing legal support for the designing lawful business models of new businesses and for funding. In addition, I have been providing legal support for businesses in the field of medical and beauty based on my knowledge of laws relating to medicals including the Japanese Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices and the Japanese Medical Care Act. In particular, I have been providing extensive support for numerous businesses operating in new field such as Software as Medical Device (SaMD) and Non-Software as a Medical Device (Non-SaMD), telemedicine/home call medical services, and also for companies utilizing medical information by employing new technologies such as AI. I am also addressing legal issues faced by companies using advance technologies. Furthermore, I have expertise in the latest marketing trend and offer advice on marketing strategies based on the Japanese Act against Unjustifiable Premiums and Misleading Representations, advertising regulations in Japan such as medical advertising regulations, and medical and pharmaceutical devices regulations.

Tsutomu Hoshiba
Kievit Scientific LLC President
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Other
For over 30 years, I've established and managed Japanese life science companies in the US and Europe. In 2021, I founded Kievit Scientific in Philadelphia, utilizing my Western-acquired experience, up-to-date information, and network. I assist Japanese companies in the pharmaceutical, medical device, and chemical sectors in their international expansion, from strategic planning to product development and market entry. Harnessing my understanding of different cultures and wide-ranging scientific knowledge, I provide solutions to challenges faced by Japanese academia and startups in their global ventures, with the aim of supporting as many Japanese companies as possible to succeed overseas. Furthermore, through these endeavors, I promote the widespread adoption of innovative medical technologies, striving to realize a society where the benefits are extended to many.

 A comprehensive portal site for Medical Innovation Support Office (MEDISO).
A comprehensive portal site for Medical Innovation Support Office (MEDISO).





