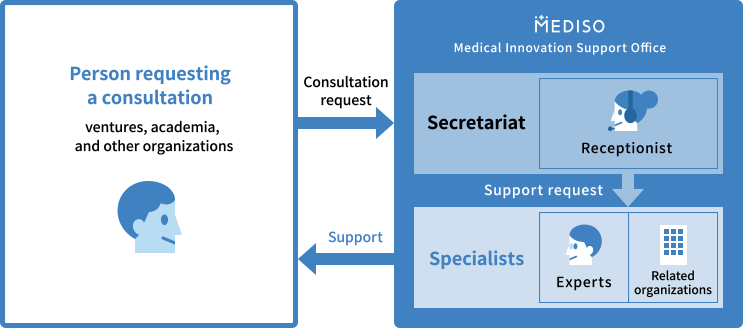
Specialists Introduction
Medical Innovation Support Office support consultants in cooperation with experts (specialists) in each field of R&D, pharmaceutical affairs and insurance, intellectual property management, management and financial accounting, marketing, legal affairs, and international development, as well as related organizations, including the Ministry of Health, Labour and Welfare.
What is Specialists
Experts in each field of R&D, pharmaceutical affairs and insurance, intellectual property management, management and financial accounting, marketing, legal affairs, and international development.
Depending on the specifics of your request, we offer support in cooperation with appropriate specialists and related organizations, including the Ministry of Health, Labour and Welfare.
Introduction of Support Providers
We will introduce registered support providers
(updated as necessary).
Search the area
Search the area
Mirai Miyahara
President of i Access Consulting Inc.
Main specialty areas
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy
Miyahara holds a Master's degree in Business Administration from the Graduate School of Economics at Tohoku University. She possesses 17 years of professional experience in Regulatory Affairs, QMS, business planning, and overseas marketing at a leading medical device company and Notified bodies in Japan. With a specific focus on multi-country regulatory compliance and extensive experience in overseas medical device markets, Miyahara has demonstrated proficiency in navigating the regulatory landscapes of diverse markets, including Japan, China, India, East Southern Asia, EU and the U.S. She has a proven track record of achieving success in supporting medical companies with regulatory compliance, facilitating international expansion, and establishing strategic partnerships with local entities. Miyahara is also an adviser for JETRO medical devices international expansion program.

Kazuhiro Ikumi
MEDICOLAB Co., Ltd.
Main specialty areas
- Medical devices
Specialized support fields
- Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management - Other
Board-certified neurologist, CEO at MEDICOLAB, Co., Ltd. My initial job was a medical doctor at a tertiary care emergency hospital. I have 14 years of clinical experience in Parkinson's disease, dementia and other neurodegenerative diseases. MEDICOLAB, Co., Ltd. receives PR and development support from Fujitsu, NVIDIA, Amazon, and Microsoft. I am involved in business development in the CNS area and support researchers for their idea and trial products or services for commercialization.

Yutaka Kono
Guest professor
Drug Discovery Science Division,Institute for Open and Transdisciplinary Research Initiatives, Osaka University
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Business
planning - Fundraising
- Management
Strategy - Other

Masaho Ishino
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Management
Strategy - Intellectual
property
management - Other
After engaging in medical research for 25 years, I became a patent attorney (JAPAN). Over the next 20 years, I have supported medical researchers in creating intellectual property and planning research strategies, as well as engaged in contract practice and technology transfer of university IP. Intellectual property strategies for regenerative medicine and other new modalities require not only a high level of expertise in both cutting-edge medical research and intellectual property rights, but also comprehensive knowledge covering clinical development and regulations. Utilizing my experience of contributing to the development research and practical application of university-originated seeds as a representative of an institution responsible for the AMED's Project of Translational and Clinical Research Core Centers and an organizer of nationwide academia intellectual property networks, I aim to contribute to raising the potential of intellectual property of academia/venture.

Eiichi Yamaguchi
Eiichi Yamaguchi, Ph.D. President A1 Partners
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy
Ph.D from Faculty of Pharmaceutical Sciences Kyushu University. Currently engaged in management and business development consulting for pharmaceutical companies, biotech ventures, and consulting firms. Over 20 years expertise of business development at Shionogi Co., Ltd., a major pharmaceutical company as well as bioventure companies. Senior Director, Business Development & Corporate Planning Dept., Shionogi. A lot of global business experience such as the first Managing Director, Shionogi Singapore, Vice President, Shionogi Qualicaps, Inc., North Carolina, and Shionogi New York. Board of Director & President & COO, a bioventure company. The first chairman, Japan Pharma Alliance Association. The Vice Chairman, Public Private Alliance Conference associated with Infectious diseases led by Dr. Shigeru Omi. Also engaged in Director, International Committee, JPMA. Last but not least, selected as Mentor for Blockbuster Tokyo 2021

Hiroya Kuwahara
Department of Neurology and Neurological Science / Institute of Innovation Advancement
Tokyo Medical and Dental University
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Management
Strategy
M.D. (graduated from Tokyo Medical and Dental University), and Ph.D. (completed doctral course at Graduate School, Tokyo Medical and Dental University). Specialist and Attending Neurologist of the Japanese Society of Neurology, and Fellow and Board Certified Member of the Japanese Society of Internal Medicine. He is engaged in 1) clinical practice, research, and education in neurology, 2) research and development of fudamental technologies for drug discovery (especialy nucleic acid drugs and drug delivery system), 3) promotion of clinical research and industry-academia collaboration, at Tokyo Medical and Dental University. He has a career of a Deputy Director at Research and Development Division, Health Policy Bureau, at the Ministry of Health, Labour, and Welfare from 2018 to 2019, and was involved in establishing a nationwide system for promoting clinical research and accelerating venture companies in the healthcare field. He provides consultations on the needs of clinical practice, on collaborative research and field provision in relation to project implementation, and on clinical research and development.

Masakazu Masujima
Partner, Mori Hamada & Matsumoto
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Fundraising
- Management
Strategy - Intellectual
property
management
Masa is a guru of startup legal advisory space in Japan. Based on the experiences at Palo Alto office of Wilson Sonsini Goodrich & Rosati, his advice is West Coast style; progressive, risk-base and proactive. Through more than 2 dacades of dedication to the startup space, he has advised IPOs, M&As and major cross-border fundraising for may startups, including biotech and healthcare ones, and have also worked with entrepreneurs through a variety of touch situation, such as disputes between founders, problems with investors as well as intellectual property disputes. Masa has served as a council member of numerous Government committees, such as Regulatory Reform Promotion Council, the Regulatory Sandbox Committee, the Digital Market Competition Council and the Industrial Structure Council. He is admitted to NY bar and Japan bar, and a licensed patent attorny in Japan.

Yoshiki Kawabata
President, MedX Inc.
Main specialty areas
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
Advantage from over 35 years of multiple company & responsibility experience in medical device industry, helping foreign companies to enter into Japanese market. Key highlights in the past achievements are : with Johnson & Johnson Mdecial Company, worked with US R&D team to develop new product which reflecting Japanese patient and physicians needs - three years resided in Cincinnati, Ohio: HeartFlow Inc., which developed the first "program device" - FFRCT, enabling non-invasive detection of FFR, applied and received regulatory approval and reimbursement as a first product and company. : in Palette Lifesciences Japan, establishment of new subsidiary for start-up company which transferred the business from exclusive distributor. Started with minimum number of direct resources and having multiple partnerships with logistics, finance and regulatory affairs, revitalized the business and achieved a 149% growth over the last year of distributor.

Nao Yoshizawa
GRiT Partners Law offices/Willsame.co.ltd/Cabinet office advisor for National bioecnomy strategy/Keio university Medical school
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
After leaving the law firm Nishimura Asahi, [the individual] established the law firm GRiT Partners and assumed the role of managing partner. They also serve as the representative of Willsame Corporation and hold qualifications as a lawyer, patent attorney, CFE (Certified Fraud Examiner), Applied Information Technology Engineer, Information Processing Security Support Engineer, and IT Strategist. They have served as a committee member for the formulation of standards in electronic commerce and related information transactions at the Ministry of Economy, Trade and Industry, as an expert in the Cabinet Office's bioecnomy strategy, and as an advisor to Biock. They have been involved in the establishment of the Japan Chapter of Aging2.0 in Silicon Valley and served as a mentor for Alchemist Accelerator. A graduate of the law faculty at Hitotsubashi University, they completed intellectual property and machine learning programs at Stanford University, as well as multiple AI programs at MIT. They serve as a program committee member for the Graduate School of Pharmaceutical Sciences at the University of Tokyo and the Medical Innovation Human Resources Development Course at Tokyo Medical and Dental University. With a deep understanding of international perspectives on digital health, they provide advisory support for startups and pharmaceutical companies' DX (Digital Transformation) programs. They founded the Life Science Incubation Council and focus on launching projects and connecting them with ecosystems both domestically and internationally.

Kiyoaki Kojima
Partner
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
Kiyo advises clients in corporate formation, governance, and compliance matters; mergers and acquisitions; joint ventures; leasing and licensing matters; distribution and franchise arrangements; and a wide range of commercial transactions.An important part of Kiyo’s practice includes his work with international companies in relocating and/or establishing ventures in the U.S. He assists companies and investors with incentives and site selection; real estate acquisitions and construction agreements; corporate formation and governance; asset purchases and joint ventures; regulatory compliance; and contracts of all types to help ensure a seamless U.S. launch or transition.He also counsels clients in virtually all areas of labor and employment law, including drafting and/or interpretation of employment agreements, company policies, and handbooks; union-avoidance training; countering discrimination and harassment claims; hire, fire and discipline issues; and dispute resolution procedures. Active in the community, Kiyo is a member of the Atlanta Bar Association, a board member of the Japan America Society of Georgia, and a board member of the Japanese Chamber of Commerce of Georgia. He also serves in “advisor” or “supporter” roles in various entities in Japan, such as InnoHub, MEDISO, and IDEC Yokohama.

Tadashi Sameshima
Main specialty areas
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy
I have been engaged in product development and business operations of medical devices and regenerative medicine products for 37 years at Terumo Corporation. I have experienced the development of medical devices such as extracorporeal circulatory and blood transfusion devices, as well as the development to commercialization of cellular products. In addition, I am able to provide specific advice on various stages of development based on my participation in joint research with universities, collaboration among companies, discussions with government agencies, and activities of industry associations.

Tomokatsu Hongo
Keio Innovation Initiative, Inc
Corporate Officer
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Business
planning - Fundraising
- Management
Strategy
Ph.D. in Biological Chemistry from Nagoya University. At Summit Pharmaceuticals International Corporation, I was engaged in VC fund management, investment and sales support for bio-ventures, and domestic sales agency business for overseas bio-ventures. In 2016, I joined Keio Innovation Initiative (KII) as an investment manager in the medical and health field. I have been investing in drug discovery, regenerative medicine, and medical device ventures by leveraging his business experience in the pharmaceutical industry and career as a researcher, and providing a wide range of hands-on support, not only in terms of funding, but also in business development.

Takahiro Mizumoto
Ant innovations Co., Ltd
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
Kyoto University Graduate School of Medicine, Master’s Program in Intellectual Property Management.
After passing the Japanese Patent Attorney Examination, gained venture investment experience in high-tech sectors including IT and healthcare at NIFS SMBC Capital (later split into Daiwa Corporate Investment and SMBC Venture Capital).
Subsequently joined Showa Shell Sekiyu, where he led the planning and development of multiple new services spanning payment solutions, retail electricity, and mobile app businesses.
With the launch of the University of Tokyo Innovation Platform (UTokyo IPC), he joined the firm and founded 1stRound, a cross-university incubation program. He later spearheaded the establishment of an approximately JPY 25 billion open-innovation promotion fund and served as its Chief Investment Officer.
In November 2025, he founded Ant Innovations, a newly established venture capital firm and subsidiary of the private equity firm Ant Capital Partners, and assumed the role of Representative Director and Partner.

Chia-Feng Lu
Shareholder(Senior Partner), Greenberg Traurig, LLP
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
Chia-Feng Lu represents life sciences companies and industry associations in strategic regulatory and legislative worldwide to guide and develop new legislation and policy, to frame effective approaches to working with the various agencies on compliance and investigation issues. He has intensive experience in provision of the guidance to companies introducing novel technology products, such as AI, block chain, 3D-printing, cancer immunotherapy, digital health, cell / gene therapy, precision medicine-related products and services, RNAi drug, as well as SMART device. In his past experience, he has already assisted companies in translating the scientific ideas from Nobel Laureates into the successful commercial launch of at least two products. In addition, he advises investment banks, private equity firms, and venture capital groups on their evaluations of the technologies, deal structures, and the resulting business impacts, as well as regulatory uncertainty with respect to novel technology and compliance. He specializes in various issues in connection with biotech investors or life sciences companies’ research & development strategy, portfolio management, corporate strategy planning, partnerships, licensing, project finance, auction, investment, and M&A. He has also served as an adjunct faculty member of a number of leading academic institutions in the U.S. and Japan, and an advisor to numerous government agencies.

Takeshi komatani
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy - Intellectual
property
management - Other
Takeshi S Komatani is currently principal litigation certified patent attorney in the field of chemical/pharma and bio/life sciences at a patent law firm, and has a number of counsels, including in academia, start-ups and big pharma internationally. Takeshi received his PhD from the University of Tokyo, Japan, received his LLB and LLM (global legal practice) from Keio University, and is currently a visiting professor at Kobe University, teaching IP strategy in entrepreneurship and Doshisha University (Kyoto). He has completed CEIPI Summer School on IP (University of Strasbourg), France. He is also qualified as a pharmacist with a certificate of Kampo and natural medicines specialist qualification. He was a researcher at F Hoffmann-La Roche in Basle, Switzerland. He has completed the EU-recognised PharmaTrain course at Osaka University, and is qualified as a board-certified member of the Japanese Association of Pharmaceutical Medicine. He is a member of the AIPPI and vice chair of TRIPS SC and a member of IP-GRTK SC and Pharma SC, and a member of the editorial board of the Pharmaceutical Patent Analyst (UK). He is also a member of JPAA, APAA, IPAJ, PSA, JPA, CSAIP and AAAS. He has lectured in academia, including at Japanese and German universities and WIPO Academy, and contributed to a number of articles.

Jun Yamadera
Eyes, JAPAN co. ltd.
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy - Other
Jun Yamadera founded Eyes, JAPAN co. ltd. in 1995, the first IT startup from the University of Aizu, Fukushima. In the past 20 years, he has been working on various cutting edge projects such as exporting Fukushima rice via web in 1995, making world’s first virtual pottery system, making CG of historical archives of national treasure of Japanese castles, temples, traditional dances and medical motions using motion capture. He is a pioneer in Augmented Reality wearable technologies, Medical x IT and has been organizing world’s first medical security hackathon since 2012. His team won the the championship in Developers Challenge 2013 Health 2.0 in Silicon Valley. He is the Health 2.0 Fukushima Chapter Leader and a TEDxKobe 2015 Speaker. He started a project called “FUKUSHIMA Wheel” in the aftermath of the terrible disaster caused by the earthquake and nuclear accident in Fukushima, JAPAN in March 11, 2011. More than 24 years experience in Cutting Edge Technologies such as AI, Blockchain and Cyber Security. He is also advisor of Safecast and LivingAnywhere.

Masatoshi Tachibana
BPM Consulting Office Representative
Main specialty areas
- Medicines
- Medical devices
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy
He worked in the medical device division of a precision equipment company for over 25 years, working in a series of tasks spanning the product life cycle, including marketing (domestic and overseas market research, business development), product planning, legal and regulatory compliance, quality management system construction. He has experience managing numerous projects, including those outside the company also. His policy in conducting business is to maximize the performance of the organization by promoting overall optimization, making decisions based on a bird's-eye view, and building good relationships with all of stakeholders. Based on these knowledge and policies, He provides fact-based and risk-based support to ventures and small and medium-sized enterprises aiming for practical application in the medical field.
-e1571192029577.jpg)
Yumi Kawata ,MD, PhD
Hedgehog MedTech, Inc.
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy
She is Founder and CEO of Hedgehog MedTech, Inc, a start-up Digital Therapeutics (DTx) company. Previously, she was a manager with SoftBank Corp, where she evaluated new Healthcare & Life Science investment opportunities, managed relationships with strategic partners. Prior to SoftBank, she was a Government Relations specialist with Medley, Inc, where she built and maintained the relationships with Ministry of Health, Labour and Welfare and Academia. She launched researches and POCs, contributing to the spread of telemedicine in Japan. She also served as a technical officer for MHLW. she was involved in the management of researches on Rare and Intractable diseases and Infectious Diseases.

Yasuhisa Matsukawa
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Management
Strategy - Intellectual
property
management

Hitoshi Takuma
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Fundraising
- Management
Strategy - Intellectual
property
management

Masashi Matsunaga
Biospire Japan Ltd. / Biospire UK Ltd.
CEO
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy
As an experienced global business development strategist, Dr Masashi Matsunaga provides business soft-landing, clinical and regulatory support to the UK and European market. He also enables to support IP management, marketing and pitch-deck brush-up in the MEDISO support package. He is based at Harwell Science & Innovation Campus, a core UK innovation hub located near Oxford.

Hidehiro Ando
President & Representative, Andy Pharma Partners Inc.
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Fundraising
- Management
Strategy
He graduated from the Department of Veterinary Medical Science, Faculty of Agriculture, University of Tokyo. He joined Tanabe Pharma Corporation (present Mitsubishi Tanabe Pharma Corporation). After being assigned to the research department of pharmacokinetics, he was involved in partnership-related business of international development department, research and development planning department, etc., including establishment of a joint venture with overseas companies. Subsequently, he worked in the technology transfer department of RIKEN (Institute of Physical and Chemical Research), a regenerative medicine venture, Bayer Yakuhin, a drug discovery venture, and he established a personal office. He has engaged in business development and licensing work for 40 years, and now, based on his experience and networks, he is supporting business development activities, such as licensing agreements and joint research of domestic and foreign pharmaceutical companies/ventures. He provides comprehensive support services ranging from looking for appropriate partners, preparing the draft of contract outline, contract negotiation, and alliance management with partners after the partnership agreement. Especially, based on experience of practicing on both sides of venture and pharmaceutical companies, he keeps in mind providing informative advice and proposals for specific action plans.

Yoshinori Shinoki
Pharma Initiative Support LLC
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy
He graduated from the Department of Biopharmaceutical Sciences, Kyoto Pharmaceutical University, and is a licensed pharmacist. At Sawai Pharmaceutical Co., Ltd., and Eli Lilly Japan K.K., after taking charge of clinical development, business planning, etc., he acquired experience in planning and implementing various clinical development plans as a project leader for newly developed items in the project management department. In addition, he led NHI price negotiation strategy, post-marketing clinical startegy, and market planning. After that, he built up clinical development department at Global CRO. he also led the planning of the clinical development for Japan for small and biological products, as well as negotiating with regulatory authorities. Recently, in addition to medicines, he also led the development of clinical development strategies of medical devices and the products of regenerative medicine (gene therapy products and cell/tissue processed products).

Kenichi Yamahara
Professor at the Laboratory of Molecular and Cellular Therapy, Institute for Advanced Medical Sciences, Hyogo Medical University
Representative Director of CTEX Co., Ltd.
Main specialty areas
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy - Intellectual
property
management
From the time of a former position at the National Cerebral and Cardiovascular Center, he planned and implemented the formulation of the amnion-derived mesenchymal stem cells and investigator-initiated clinical trials for acute grafts versus host disease/Crohn's disease under the support by Health and Labour Sciences Research Grants/AMED. He often had PMDA consultations on the products of regenerative medicine and acquired knowledge of laws and regulations. Taking advantage of the experience, he founded a venture, Japan Biomedical Co., Ltd., which manufactures the domestic bovine serum NeoSERA® for the products of regenerative medicine by satisfying the standards for biological materials in January 2017. Furthermore, in order to aggressively develop investigator-initiated clinical trials of cell therapy making use of university hospital infrastructures, he founded a venture, CTEX Co., Ltd., from Hyogo Medical University in February 2018. Recently, he participated as a medical expert in the company developing the regenerative medical product and is a principal investigator for investigator-initiated clinical trials for the development of cell therapy in the medical device category (adopted by AMED). Currently, he is working on the practice of cell therapy development initiated at the university hospital while concurrently serving in the basic and clinical departments of Hyogo Medical University.

Atsushi Usami
The University of Tokyo Edge Capital Partners Co., Ltd. (UTEC)
Partner and Board Director
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
He holds a Ph.D. in pharmaceutical sciences from the University of Tokyo and is a licensed pharmacist. Prior to joining UTEC in October 2013, he worked as a consultant at Mitsubishi Research Institute. Currently serving as a Partner and Board Director, he oversees venture investments and provides management support primarily in the life sciences sector for seed and early-stage startups. He has played a key role in investments in companies such as OriCiro Genomics, Inc. (acquired by Moderna, Inc.) and Repertoire Genesis, Inc. (acquired by Eurofins Scientific SE). In recognition of his contributions, he was awarded the Encouragement Award for Venture Capitalists at the 23rd Japan Venture Awards in 2023.

Kazushi Kamitani
JPRO INCUBATION OFFICE, FOUNDER
Main specialty areas
- Medicines
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management - Other
He worked for domestic & international life science firms in the areas of product development, marketing, PR&IR, international business, and business development.
After serving the US subsidiary of a Japanese firm as a board director & CFO and the European startup as CEO, he established the incubation office.
He supports oversea startups to explore Japan market from business & administration side, providing info, advisory and functional services.
International MBA, Certified Specialist of Intellectual Property Management, Financial Planner, IT Coordinator

Daisuke Sugiyama
Hiroshima University, Translational Research Center, Professor
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Fundraising
- Management
Strategy - Intellectual
property
management - Other
He obtained his PhD thesis at University of Tokyo. After working at several hospitals as a clinician, he has engaged in medical reserach at University Paris 6 (French Government Scholar), Dartmouth College (JSPS research fellow), Kyushu University and Hiroshima University. Based on the experiences working at ARO research core hospital and starting up bio-venture company, he supports and conducts both translational and clinical researches.

Miyuki Shimizu
Medical Lab Partners Corporation
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy - Intellectual
property
management
She graduated from the Graduate School of Natural Science, Chiba University. She holds the degrees of doctor of medicine and master of management information. She worked on the development of medical devices and pharmaceutical products in a wide range of fields related to transfusions, surgical operations, general-purpose medical devices, dialysis, and rehabilitation at Terumo Corporation. She also has experience in a series of project developments from planning a project to commercializing at a medical practice, as well as experience in management. Since 2015, she has provided consulting services to more than 350 companies entering the medical industry as her current role. She is trying to provide consulting services with the viewpoint of the individual company in mind.

Satoshi Fukushima
Globis Capital Partners & Co.
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy
He graduated from the Faculty of Economics, University of Tokyo. He was involved in investment activities in reformed fields of existing industries, including digital health, at Globis Capital Partners & Co. (GCP). He served as an outside director of several companies, including Medley, Inc., Kakehashi Inc., and Yoriso. Co. Ltd., and provided support to their management personnel mainly in strategy formulation, development of the organization, and finance. In addition to investigation activities, he provided support for fund structuring and open innovations provided by large companies in healthcare fields.
Before joining GCP, he was serving as an M&A advisor mainly in the technology, media, and telecommunication fields and worked on financing at the general headquarters of German Securities Finance Corporation.

Masakatsu Noguchi
SMILE CURVE, Inc.
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Management
Strategy
Ph.D. in life sciences. Worked at consulting firm Dream Incubator, consulting in industry-academia collaboration, business support for tech startups (bio, semiconductor, environmental energy), new business development for large corporations, and overseeing government mega-projects. Previously employed at Abbott Japan and Singapore, managing product marketing for diagnostic tests, pharmaceuticals (women's health), and Asia-Pacific marketing for medical IT. As Business Development Director at Sanamedi, facilitated the commercialization of new medical devices, handled regulatory applications and market launches, and supported investments in and assistance to startups. Additionally served as an evaluator for AMED, and as a mentor for NEDO and university accelerator programs. Currently working independently, he serves as a specially appointed associate professor in the Hiroshima University and as an strategic advisor of MEJ.

Hiroaki Kato
Visiting Professor, The Graduate School of the Digital Hollywood University
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy
He is a former government official of the Ministry of Health, Labour and Welfare and is working as a doctor specialized in digital health, including telemedicine, AI, and IoT. As an ophthalmologist, he has experience in more than 1,500 surgical operations. He developed instruments for cataract operations and ophthalmic consultation services. He is a rare person who has experience in three areas—medical practice, medical systems, and business—which are required for medical care and the healthcare business, and he understands these areas, providing support for the development of new business covering the full range of medical care. He is also familiar with Japanese medical venture companies and works as a consultant, advisor, and director for large enterprises and venture companies. He wrote 41 books, including Medical Care 4.0 — Medical Care in the Fourth Industrial Revolution Era—.

Ayuko Nemoto
Aquaxis Law Office
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Fundraising
- Management
Strategy - Intellectual
property
management
My practice focuses on legal and compliance matters related to healthcare and life science companies. I have been advising on healthcare related laws and regulations, working on legal due diligence for investment, M&A, contract negotiation, and all the other general corporate matters (including domestic and international corporate matters, intellectual properties, labour laws, M&A, JV, IPO support, contract review, personal data, pharmaceutical and advertisement regulations, commercial dispute, accident, communication with relevant authority). Also act as an Advisor of the International Affairs Working Group and MA & Clinical Trial Working Group of Japan Pharmaceutical Industry Legal Affairs Association, Supporter of HealthcareInnovationHub held by Ministry of Economy, Trade and Industry and a member of IRB of the Council for Industrial use of Biological and Environmental Repositories (CIBER). Previously at international law firms.

Nobutaka Tani
Chair Person, Nonprofit Organization Japan Organoid Repository
Independent Consultant (New Business in Life Sciences)
Main specialty areas
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Business
planning - Management
Strategy - Intellectual
property
management
He graduated with a master’s degree from the Department of Synthetic Chemistry, Graduate School of Engineering, University of Tokyo. He led the research and development and the launching new project for medical devices, including blood purification system and brain and heart catheters, at Kaneka Corporation. After that, he was involved in RD of the whole life science, including biopharmaceuticals, as the head of the Life Science RD Center, while investing in medical venture companies such as DDS and establishing joint ventures. He then moved to JSR Corporation and established JSR Life Sciences. He led the launch of the life science project for M&A as the first president since JSR Life Sciences was established. As a part of open innovation, he established a CVC and invested in or acquired venture companies in Japan and overseas, while successively holding the positions of president of the invested companies.

Makoto Shigematsu
Representative Director, Kai Fostering Partners Inc.
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
He worked on a new project research and development, trial manufacture, mass production, and commercializing of glass delay line element for VTR at Asahi Glass Co., Ltd. He moved to CSKVC by making use of his relevant experience. As a venture capitalist, he engaged mainly in establishing, financing, fostering, and managing venture companies. He practiced monetizing of novel technologies, timely financing, establishing organization and management, sharing an exit strategy as a milestone, active use of intellectual property rights (open and close), and business talks and negotiations (not bargaining) hated by engineers. He has providing support for company’s growth fostered by competition and cooperation, while being employed by traditional companies for 20 years and by venture companies for 20 years with an understanding exactly the opposite of the corporate culture of both.

Tamaki Nara
Lecturer, Medical Technology Innovation Center, Juntendo University
Director, Orbis Planning Co., Ltd.
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy
Master of Business Administration(MBA). Field of expertise is innovation management. While working at a major precision equipment manufacturer, engaged in new business, product planning, and corporate planning. Mainly engaged in commercialization in the medical equipment and healthcare field (such as Medical IT), as well as the establishment and management of internal joint ventures. During this time, I became acutely aware of the great significance of building an appropriate collaborative development system between medical researchers and companies, and the necessity of a place for co-creation for both parties to work together. Since then, I have been involved in providing guidance on the implementation of research and development in the medical field and building plans for industry-academia collaboration, leveraging corporate technology and academia's knowledge at medical graduate schools and hospitals. Also have been supporting Start-ups mainly from academia.

Jun Utsumi
CEO, TIR Research Consulting LLC.
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy - Intellectual
property
management
JU graduated from the Graduate School of Veterinary Medicine, Hokkaido University. He holds DVM, D.Sc., MBA and has licenses of Professional Engineer (biotechnology), Radiation Protection Supervisor and Certified Scientist of Medical AI. He was engaged in research and clinical development at Toray Industries, Inc. and succeeded in commercializing the world's first opioid kappa agonist (received Awards from the Pharmaceutical Society of Japan and Okochi Memorial Foundation). After the business career, he served as a professor in Hokkaido University and Kyoto University, as well as an expert in Pharmaceuticals and Medical Devices Agency (PMDA) and a senior consultant for intellectual property at the Japan Agency for Medical Research and Development (AMED). Over 30 years of experience led him to establish a consulting company in 2018, he is also involved in medical DX support and won the Japan Open Innovation Award in 2023. He is part-time lecturers at the graduate schools of the University of Tokyo, Kyoto University, Tokyo Medical and Dental University, and the University of Tsukuba. He published “A Guide to Integrated Strategy in Drug Discovery” (Nanzando Co., 2015).

Yasuo Sasaki
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy - Intellectual
property
management
He graduated with a doctoral degree in science from the Department of Chemistry, Graduate School of Science, Hokkaido University. He was involved in the research and development of pharmaceutical products as an employee of Asahi Kasei Pharma Corporation. He was mainly in charge of preclinical studies and regulatory affairs. From 2013, he provided support for research and development by academia, drug discovery venture companies, and small and medium enterprises centering on the eastern part of Shizuoka Prefecture. Since 2018, he has been providing support services as a freelance advisor or coordinator in the life science field.

Satoshi Ogawa
TMI Associates Partner (Attorney)
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Fundraising
- Management
Strategy - Intellectual
property
management - Other
Satoshi Ogawa, Ph.D., is a partner at TMI Associates specializing in life sciences and healthcare. After earning his Ph.D. in Life Sciences from Kyoto University, he joined TMI Associates as an attorney. Following a three-year secondment at a law firm in New Delhi, India, he has been based at TMI’s Kyoto Office since 2019. Dr. Ogawa advises both Japanese and international clients on cutting-edge technologies, including regenerative medicine, digital health, and AI-driven healthcare solutions. His work covers startup support, patent licensing, industry–academia collaboration, regulatory compliance, and intellectual property disputes. He works extensively with universities, startups, pharmaceutical and medical device companies, venture capital firms, and public institutions. He also serves as an advisor to BioCommunity Kansai (BiocK) and HVC KYOTO, and contributes to conflict-of-interest (COI) committees and institutional review boards (IRBs). Dr. Ogawa is passionate about advancing innovation and helping clients navigate complex legal and regulatory challenges in the life science ecosystem. His publications include “Startup Intellectual Property and Legal Guidebook – Key Points for Early-Stage Bio and Life Science Ventures” (METI-Kansai) and “Healthcare Business Legal Consultation Handbook” (Chuokeizai-sha).

Kumi Sakurai
早稲田大学大学院理工学研究科で生体工学を専攻後、慶應義塾大学病院で10年以上にわたり血液研究に従事。その後、外資系医療機器メーカーであるベックマンコールター(2005年~)、日本メドトロニック(2007年~)、セントジュードメディカル(現アボットメディカル、2015年~)のマーケティング部門にて、医療機器の市場導入や事業戦略を担当。2019年、医療機器・ヘルスケア機器の開発支援を専門とするインキュベーター、プレモパートナー株式会社を共同創業。研究者・スタートアップ・大企業を対象に、事業戦略策定から薬事・保険償還・上市までを一気通貫で支援し、国内外のネットワークを活かして医療機器の事業化を推進している。
文部科学省 科学技術・学術審議会専門委員、東京都AMDAPカタライザー
早稲田大学大学院修了(工学修士)。金沢工業大学大学院修了(MBA)
Main specialty areas
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Management
Strategy
After earning a master’s degree in Biomedical Engineering from Waseda University, Kumi Sakurai engaged in over ten years of hematology research at Keio University Hospital. She then transitioned to the medical device industry, joining Beckman Coulter in 2005, Medtronic Japan in 2007, and St. Jude Medical (now Abbott Medical) in 2015, where she worked in marketing and business strategy for innovative medical technologies. In 2019, she co-founded Premo Partners Inc., an incubator specializing in the development and commercialization of medical and healthcare devices. Premo Partners supports researchers, startups, and large corporations throughout the entire innovation process—from business strategy formulation, market analysis, and regulatory and reimbursement planning to commercialization. Leveraging extensive domestic and international networks, the company provides end-to-end support to accelerate the delivery of essential medical technologies to patients and healthcare professionals. Through its comprehensive expertise in regulatory pathways, evidence generation, and global market access, Premo Partners plays a vital role in fostering Japan’s MedTech ecosystem and bridging it with the global healthcare innovation community.

Saho Murakami
VISIONEO LLC CEO
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Business
planning - Fundraising
- Management
Strategy
After gaining clinical experience as an ophthalmologist, I served at the Pharmaceuticals and Medical Devices Agency (PMDA), where I was responsible for reviewing and providing consultations on pharmaceuticals, medical devices, and regenerative medical products in the fields of CNS, sensory organs, and anesthesiology. My work included designation reviews for Sakigake (pioneering) and orphan drugs, as well as post-marketing safety activities. Currently, I support business and clinical development across the healthcare and life sciences sectors, offering strategic and R&D advisory services to major corporations, startups, and venture capital firms.

Tetsuo Miura
Specially Appointed Researcher, Center for Clinical Sciences, National Center for Global Health and Medicine
Main specialty areas
- Medicines
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Management
Strategy - Other
I worked as a product and business development manager primarily in the fields of diagnostic equipment-reagent systems and medical devices at major foreign pharmaceutical and domestic medical device companies, and also participated in global product development teams at the US and French headquarters. I have medical science knowledge in the fields of infectious diseases, immunology, cardiovascular diseases, metabolism, reproductive medicine, and laboratory medicine, as well as information on the medical field. After mandatory retirement, I served as a senior researcher at the National Center for Global Health and Medicine, responsible for developing and implementing international collaborative clinical research and clinical trials for pharmaceuticals and medical devices with clinical trial networks in Southeast Asian countries. Currently, as part of an industrial-medical collaboration project, I also provide guidance and support to companies in product development with an eye toward exit strategies such as obtaining pharmaceutical or WHO PQ approval, and being included in guidelines in cooperation with related medical sectors and societies.

Aiko Kato Sullenberger
RykoTECH President
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy
Aiko Kato Sullenberger, CEO of RykoTECH, a significant contributor within the Boston startupecosystem for several years, brings a wealth of experience in various roles, spanning investor,startup, and accelerator. She provides specialized advisory services comprehensive strategicconsultation and facilitation of market entry into the US across diverse sectors, including drugdiscovery, medical devices, digital health, digital therapeutics and biotech. Aiko can help • Fundraising • Strategic Partnership Development • Create Deal Flow (Connecting with Potential Investors) • Identifying Partners and Advisors • Assistance in Facilitating Clinical Trials • Corporate Strategy: Investment Thesis, Portfolio Analysis • Market / Trend Research & Analysis • Go-to-Market Strategy • Academic Business Liaison: Interdisciplinary Project Lead • Cross-Functional Team Building

Tomoyo Mori
Fast Track Initiative, Inc. Capitalist
Main specialty areas
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Fundraising
- Management
Strategy - Other
Tomoyo obtained her medical license and began her career by engaging in training and clinical practice in medically underserved areas. She then joined the Department of Cardiovascular Medicine at Tokyo Women's Medical University Hospital, where she treated patients with heart failure, heart transplants, and myocardial infarctions. At the university hospital, she also participated in clinical trials for medical device startups. In parallel with her clinical work, she was involved in domestic and international business development for medical device startups and led a team while participating in U.S.-based MedTech accelerators (MedTech Innovator, Fogarty Innovation). She holds an MBA from the University of Michigan's Ross School of Business and is a board-certified internist by the Japanese Medical Specialty Board as well as a certified occupational physician by the Japan Medical Association. Currently, she is part of Fast Track Initiative, where she focuses on investment, company creation, and business development for medical device startups.

Kazuhiro Terashima
CaTe inc. CEO
Fujita Health University, Department of Cardiology, Assistant Professor
Main specialty areas
- Medical devices
Specialized support fields
- Business
planning - Fundraising
- Management
Strategy - Other
Cardiologist, Fellow of Cardiovascular Intervention and Therapeutics, Registered Cardiac Rehabilitation Instructor, Certified Occupational Health Physician and Clinical Education Instructor. He graduated from Nagoya University School of Medicine in 2011. After working at the Japanese Red Cross Aichi Medical Center, Nagoya Daiichi Hospital, Sakakibara Memorial Hospital and other institutions, he joined Fujita Health University in 2023 as Assistant Professor of Cardiology and CCU Instructor. In 2020, he founded CaTe Inc. to research and develop programmed medical devices for cardiac rehabilitation, and to conduct clinical research and company-led clinical trials of such devices. He is currently the principal investigator for basic research on the Exercise Load Optimization function, a promising technology seed. In 2023 he closed Series A funding with JAFCO etc. as lead investor. While continuing his clinical work, he serves as CEO of the company and advises and supports other clinicians in the social implementation of clinical technology seeds.

Miyuki Nagao
Globizz Japan Corporation Tokyo Office Director
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy
After working at U.S. headquarters as an assistant for FDA registrations, FDA filings, and regulatory investigations for medical devices, pharmaceuticals, and food products, she moved to GLOBITS' Tokyo office to work as a project coordinator. While proposing and coordinating projects related to Japanese companies' entry into the U.S. market, she also provides support for attending and responding to FDA inspections in various fields, advises on FDA registration, conducts brief investigations, and teaches seminars.

Mineki Takechi
Cardio Intelligence Inc. Chief Development Officer
Director
Main specialty areas
- Medical devices
Specialized support fields
- Business
planning - Management
Strategy - Other
After working at Fujitsu Ltd. on the development of products and services such as image processing system and cloud information service for home healthcare, as well as research and development in information retrieval and AI, and new business development, I engaged in the construction of a pulmonary hypertension patient registry at the International University of Health and Welfare Mita Hospital. Since 2020, I have been serving as CDO at Cardio Intelligence Inc., responsible for the development of software medical devices for arrhythmia analysis and regulatory affairs. Since 2021, I have also been serving as COO, overseeing overall business planning and execution.

keiji Asada
Amenichi Consulting, LLC, CEO
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy - Other
Graduated from Auburn University with a degree in Chemical Engineering. Currently, serving as the CEO of Amenichi Consulting, LLC, a consulting firm specializing in regulatory compliance (FDA) and marketing support for companies entering the U.S. market, particularly in the fields of pharmaceuticals, medical devices, and regenerative medicine products. With extensive industry experience and expertise, he assists clients in developing business plans, securing funding, and establishing operational structures. Additionally, he provides comprehensive support, from the formulation to the execution of business strategies, to help clients enhance their competitiveness. Furthermore, he leverages his network to match clients with the most suitable business partners. With a global perspective, he offers customized services tailored to the needs of each client, aiming for sustained growth and success. Amenichi was established in 2018 and has supported over 50 companies since its inception.

Ruka Takeuchi
Discovery Biotech Consulting Representative
PARKS Commercialization Producer
Main specialty areas
- Medical devices
Specialized support fields
- Marketing
- Business
planning - Management
Strategy - Other
I have been engaged in marketing and business development for more than 30 years in the life science market, mainly in the medical and pharmaceutical fields, both in Japan and overseas. Currently, I have established my own consulting firm and provide business consulting services to foreign and domestic companies from start-ups to small and medium-sized enterprises. I would be happy to be of service to you by drawing on my past business experience as well as my daily experience in providing hands-on support to various companies and projects.

Saaya Hirai
Globizz Japan Corporation Osaka Office Director
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Management
Strategy
After studying abroad in New Zealand during junior high school, she became interested in doing business with foreign countries, and after graduating from high school, she entered a university in Taiwan in English and Mandarin Chinese. At university, she majored in international business administration. During her studies, she experienced corporate internships in the U.S. and Taiwan, gaining insight into the business world from her student days. Currently, as a project controller at headquarters, she is in charge of overall project management and strategic planning for companies entering the U.S. market as a project coordinator.

Teppei Konishi
Biomy Inc. CEO
Main specialty areas
- Medical devices
Specialized support fields
- Laws and
regulations - Business
planning - Fundraising
- Management
Strategy - Other
He completed his graduate studies at the Graduate School of Engineering Science, Osaka University. At NTT DOCOMO, he specialized in big data analysis and AI-driven video analysis research, contributing to new business development. After leaving NTT DOCOMO, he served as CTO at an IT venture before founding Biomy Co., Ltd. There, he led the development of a personalized medical platform utilizing AI technology, collaborating with domestic and international pharmaceutical companies and research institutions. He also secured a Class II medical device manufacturing and sales license.

Akiyo Inoko Hewett
Attorney, Smith, Gambrell & Russell, LLP
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
Akiyo Inoko Hewett is a bilingual and double-licensed attorney based in Atlanta, Georgia, United States. She graduated from Tokyo University of Foreign Studies and earned her J.D. at Keio Law School. After passing the Japanese bar exam and completing her legal training in Kushiro, Hokkaido, Akiyo relocated to the United States. She earned an LL.M. from Emory University School of Law in Atlanta, Georgia. Currently, Akiyo serves as an attorney at Smith, Gambrell & Russell, LLP, where she provides legal services to Japanese companies doing business in the United States and supports clients across a broad range of corporate matters. Her practice areas include international and domestic commercial transactions, regulartory compliance, healthcare law, and immigration law.

Kensuke Suzuki
Nagashima Ohno & Tsunematsu
Partner, Attorney-at-law
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Business
planning - Fundraising
- Management
Strategy - Intellectual
property
management
Kensuke Suzuki is a partner at Nagashima Ohno & Tsunematsu. He represents both domestic and international clients, with a special focus on cross-border transactions, with respect to pharmaceutical, medical and healthcare business, M&A and corporate transactions, fund formation and management, and financial regulations. With expert knowledge of the Japanese medical, pharmaceutical, medical device, and other various healthcare-related regulations, he advises on a wide variety of business activities in the broad healthcare sector, such as regulatory compliance, data protection, mergers and acquisitions, alliances, business startups, financing, licensing and other commercial transactions. He was admitted to the Japan Bar in 2000 and worked as visiting attorney at Kirkland & Ellis (Chicago) from 2006 to 2007. He served as a special adviser to the Ministry of Health, Labour and Welfare from 2014 to 2015. He is fluent in Japanese and English.

Hiroyuki Hasegawa
Director of Life Science, Mitsubishi UFJ Capital Co., Ltd.
Main specialty areas
- Medicines
- Medical devices
- Regenerative medicinal products
Specialized support fields
- Laws and
regulations - Marketing
- Business
planning - Fundraising
- Management
Strategy
He graduated with a master’s degree from the Faculty of Pharmaceutical Sciences, Hokkaido University. In 1994, he joined Daiichi Seiyaku Company (current Daiichi Sankyo Company, Limited) and was in charge of the areas of infectious diseases and cancers in the Department of Post-marketing Surveillance. In 2004, he joined UFJ Capital Co., Ltd. (current Mitsubishi UFJ Capital Co., Ltd.), where he served as an analyst and capitalist. From 2013, he made an attempt to use open innovation for the Development of Emerging Technologies (OiDE) Fund for the purpose of nurturing outcomes from academia-launched research to become the fundamental technology for drug discovery in collaboration with Daiichi Sankyo Company Limited(liquidated in 2023).Since the first fund in 2017, we have been promoting investment activities using a total of 50 billion yen, including the "Mitsubishi UFJ Life Science Fund No. 4" (20 billion yen).Currently, he holds the position of outside director at Kamuipharma, GAIA Biomedicine, and Luxonus Co. Ltd., as well as serving as a fellow for collaboration between industry and academia at Kyoto University Medical Science and Business Liaison Organization.

 A comprehensive portal site for Medical Innovation Support Office (MEDISO).
A comprehensive portal site for Medical Innovation Support Office (MEDISO).





